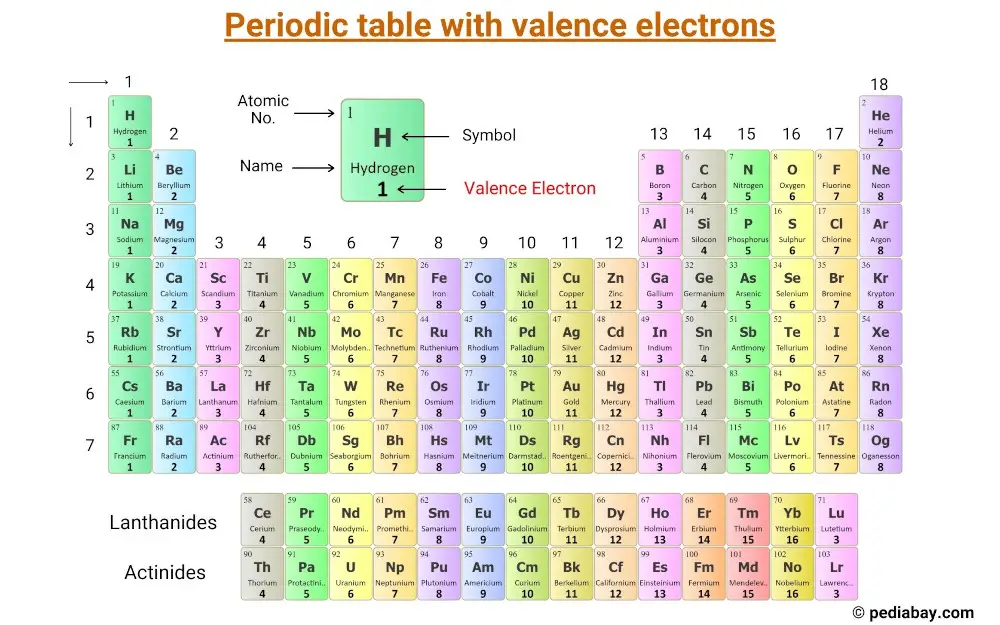
They determine how "willing" the elements are to bond with each other to form new compounds. 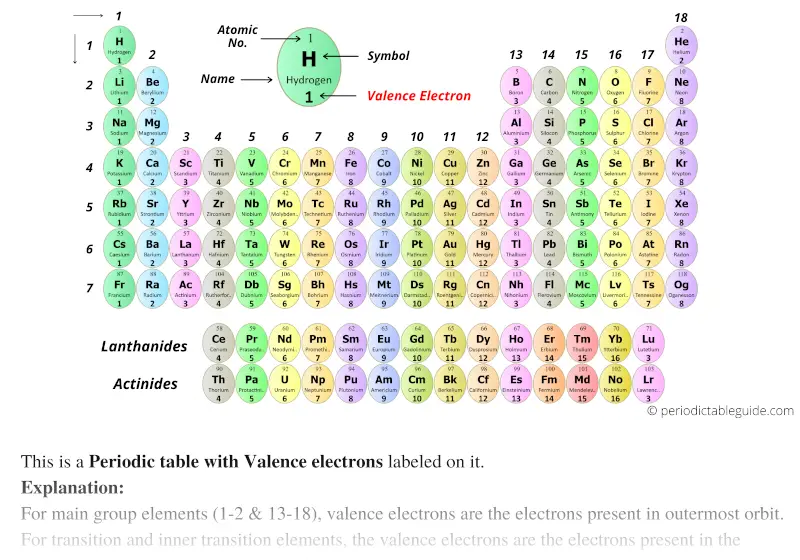
Valence electrons are responsible for the reactivity of an element. You can easily determine the number of valence electrons an atom can have by looking at its Group in the periodic table.įor example, atoms in Groups 1 and 2 have 1 and 2 valence electrons, respectively.Ītoms in Groups 13 and 18 have 3 and 8 valence electrons, respectively. Valence electrons are the electrons present in the outermost shell of an atom. Here is a video which discusses how to draw Lewis structures for atoms showing their number of valence electrons. In summary, valence electrons determine the bonding patterns of atoms. These ions attract one another and form an ionic bond. It is energetically favorable for lithium to lose one electron which is gained by fluorine.Īs a consequence, lithium acquires a + 1 charge, while fluorine acquires a -1 charge. The total of valence electrons in the second energy level for this atom is 7 (2+ 5). The highest energy level for fluorine is 2 and this energy, it has 2 electrons in the s orbital and 5 electrons in the p orbital. Since the highest energy level for lithium is 2 and it contains one electron, the valence number for lithium is one.įluorine has a configuration of #1s^2 2s^2 2p^5#. The electron configuration for lithium is #1s^2 2s^1# The alkali metals have one valence electron in their highest energy level. Valence electrons are the outermost electrons and are therefore at the highest energy level.īecause they are the outermost energy levels, they are available to participate in chemical bonding, either ionic or covalent.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
